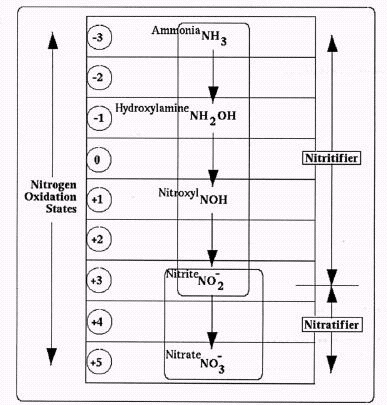
 Figure 1. Conventional Nitrification Pathway
Figure 1. Conventional Nitrification Pathway James E. Alleman, Ph.D. and Kurt Preston
School of Civil Engineering
Purdue University
West Lafayette, Indiana
Hidden literally in the shadows of every successful aquaculture operation lies a reclusive bacterial clan whose lifestyle spans the extremes of bizarre and beautiful. Few humans will ever see these magnificent creatures, miniature residents of life's trailing edge, yet without their help your fish would be downright miserable if not altogether lifeless.
The group, collectively known as nitrifiers, actually includes two discrete microbial partners tied faithfully to a life of biochemical harmony. Together, they play a critical role in every earthly ecosystem, both aquatic and soil, scavenging potentially toxic nitrogen compounds from their surroundings, including: ammonia (NH3) and nitrite (NO2-). Linked in `bucket-brigade' fashion, this nitrifying family successively transforms these nitrogenous chemicals through a delicate set of biochemical oxidations, eventually producing a soluble nitrate (NO3-) end-product.
Given their subtle and minute nature, the role of nitrifying bacteria in these reactions escaped detection until late in the nineteenth century. However, mankind had long been fascinated with nitrifications's mineralized crystalline product (i.e., nitre or saltpeter). Over a period extending nearly two millenia, ancient philosophers, alchemical wizards, and modern chemists alike tried desperately to find this enigmatic crystal's true source, driven largely by the explosive discovery of gunpowder.
Oblivious to nitre's bacterial origin, let alone its true composition, this potent crystal seemingly coalesced from unseen atmospheric spirits, an earthly offspring magically impregnated with nature's thunderous `menstruum.' In fact, nitrogen's own name (a.k.a. nitre-genesis) bears witness to this original hypothesis, forever perpetuating the quaint legacy of nitre's supposed aerial origin.
Our current understanding of these bacteria has improved considerably, though, over the past century. Shortly after discovering its biochemical source, researchers identified two separate bacterial companions responsible for nitrification. Each partner critically depends on the other, and each finds nourishment in seemingly barren substrates. Both of these bacteria qualify as lithotrophic microbes (translated from Greek, `rock eaters'), feeding on chemicals which no other bacteria would possibly use as their primary energy source.
Aside from this particularly spartan diet, using chemicals often regarded as potential toxins, their intermediate and final products are similarly unusual. Perhaps most notably, the lead organism actually makes, and then reingests, a dangerous mutagenic substance (hydroxylamine, NH2OH) whose noxious character hardly seems commensurate with normally healthy metabolic activity.
Based on our overall perception of its unusual lifestyle, therefore, the conventional wisdom for nitrifiers provided in most textbooks describes a fragile, highly sensitive life form. However, evidence collected over the past few years strongly refutes this image. In fact, recent discoveries about these unusual microorganisms suggest a far different behavior, as robust, metabolically agile microorganisms.
This article will consequently explore our current knowledge of nitrifying bacteria, detailing their metabolic behavior, qualifying their environmental likes and dislikes, and highlighting recent insights on their unique lifestyle. In addition, a set of recommended guidelines will be provided to enhance and promote their successful use within aquaculture systems.
The nitrifying bacterial clan includes two distinctly different subsets, based on their consumption of either ammonia or nitrite. Table 1 provides a breakdown of these affiliated bacterial genus and species members, covering a total of eight separate nitrifying bacteria, including: five species of ammonia-oxidizers (often called `nitritifiers'), and three nitrite-oxidizers (`nitratifiers').
Taxonomic Preferred Environs Motility Potential
Group Fresh Saline Soil Motile Non-Motile
Water Water and Mud
------------------------------------------------------------------------
Nitritifiers
Nitrosomonas europaea * * * *
Nitrospira briensis * * * *
Nitrosococcus nitrosus * * *
Nitrosococcus oceanus * * *
Nitrosolobus multiformis * *
-------------------------------------------------------------------------
Nitratifiers
Nitrobacter winogradsky * * * possibly in *
continuous
culture
Nitrospina gracilis * *
Nitrococcus mobilis * *
-------------------------------------------------------------------------
Ironically, studies recently completed (Wood 1986) on the genetic makeup of these respective subsets do not show strong evidence of a common genetic structure between these 'nitritifier' and 'nitratifier' subsets. Accordingly, each appears to have evolved from a distinctly different ancestral lineage.
On the other hand, the ammonia-oxidizing 'nitritifiers' actually appear to have much more in common with an aerobic heterotrophic (i.e., organisms who subsist on organic carbon) bacteria with a similarly fastidious diet, methane. This latter group is generically referred to as methanotrophs (i.e., methane oxidizers). Their metabolic correlation has been reinforced with five supporting observations: 1) similarities in the structure of their genetic coding (i.e., DNA composition), 2) similarities in their substrate-oxidizing monooxygenase enzymes, 3) similarities in the structure of their cell membranes, each being extensively intruded or corrugated, 4) apparent similarities in their desired substrate forms (e.g., both consuming unionized forms having molecular weights of either 16 or 17),and 5) the fact that `nitritifiers' can co-metabolize the methanotroph's substrate, methane.
After a full century of intensive study, modern microbiologists still do not fully understand just how the nitrification sequence first begins. Specifically, there is lingering confusion over the `nitritifiers' initial substrate form.
Most textbooks indicate that nitrification starts from ionized ammonium-nitrogen, NH4+-N. Suzuki's (Suzuki, et al., 1974) work, however,strongly suggested that the lead `nitritifying' organism used unionized ammonia, NH3, in a gaseous state, based on an analysis of the organism's response to elevated substrate levels. Furthermore, this behavior appears consistent with its genetic `methanotrophic' cousin, who similarly ingest gaseous methane.
Assuming that ammonia is, in fact, the starting point, Figure 1 provides a complete overview of the currently accepted nitrogen transitions through a normal nitrification sequence, covering both ammonia and nitrite oxidizers. These biochemical reactions are all respirative, with oxygen used by the organisms as a sink for electrons drawn from their reduced nitrogen substrates.
 Figure 1. Conventional Nitrification Pathway
Figure 1. Conventional Nitrification Pathway Admittedly, though, the chemical intermediates found along this sequence seldom appear at measurable or significant levels. For example, intermediate nitrite levels rarely reach the milligram per liter range (Alleman, 1985). In turn, the entire process could usually be considered a single-step jump, from NH3 to NO3-.
Under certain conditions, however, it is conceivable that the complete nitrification reaction could become disrupted, with elevated levels developing for one or more of the intermediates. A variety of stress conditions might promote this type of disruption, including oxygen deprivation or toxin presence. These factors, and their resultant impacts,
will be discussed in subsequent sections of this paper.Nitrifiers and heterotrophs maintain distinctly different growth and substrate uptake patterns. As with all bacteria, their kinetic rates depend on substrate availability. At maximal activity levels, both are able to consume several times their body weight in substrates each day. Nitrifiers, though, convert far less of their consumed substrate into new cell mass. The latter characteristic is quantified on the basis of cellular yield, which for nitrifiers is considerably less than that of heterotrophs.
Table 2 provides an overview of the generally accepted kinetic coefficients for each of these groups, including: `nitritifiers', `nitratifiers', and heterotrophs.
TABLE 2. Kinetic Characteristics of Nitrifying versus Heterotrophic Bacteria.
(see Sharma and Ahlert, 1977 and Gee et al., 1990)
Bacterial Specific Substrate Specific Growth Yield Saturation
Group Uptake Constant
(mg/mg-cells-day) (mg/mg-cells-day) (mg/mg) (mg/L)
---------------------------------------------------------------------
Nitritifiers 6-->20 0.5-->2.0 0.05-->0.08 1
Nitratifiers 10-->40 0.2-->1.0 0.02-->0.05 1
---------------------------------------------------------------------
Heterotrophs 20-->30 10-->15 0.4-->0.5 75-->125
---------------------------------------------------------------------
Another important characteristic is that nitrifiers are able to shift into their maximal activity range at a distinctly lower substrate concentration (i.e., at approximately one percent the concentration required by heterotrophs). For nitrifiers, this characteristic might be considered analogous to a `light switch' phenomenon, working either full-on or full-off, without much of a metabolic intermediate.
Many of the nitrifying bacteria use whip-like flagella to propel themselves through water, akin to an oar which drives a Venetian gondola. However, the environmental engineering literature often suggests that nitrifiers prefer a sedentary, stationary existence, attached to a solid surface rather than freely swimming. In fact, nitrifying reactor systems inherently rely on this adhesive preference. Nitrifiers could only be retained inside process reactors when attached either to a surface biofilm or as part of settleable activated sludge floc.
For most bacteria (i.e., heterotrophs), attachment stems from their exterior coating with a sticky, exocellular polysaccharide, typically referred to as `slime.' However, in the case of nitrifiers, we simply do not know if they have this sort of adhesive coat.
Recent observations, though, strongly suggest that their attachment does not involve this sort of surface slime. Indeed, nitrifiers attached to one another in dense clusters have been seen to rapidly detach in pursuit of a free-swimming migration. If slime had been the agent for attachment, it is quite unlikely that these organisms could have mustered enough energy to pull away nearly so quickly, if at all.
Aside from the inherent mechanism for attachment, another uncertainty exists as to the rationale behind their personal preference for one mode over another, either free-swimming or attached. Unstressed nitrifiers apparently prefer a free-swimming lifestyle, perhaps endowed with sufficient energy to pursue the luxury of independent locomotion (Hyman, 1991). As environmental stress (e.g., abnormal environmental conditions, lack of substrate availability, predator interaction, etc.) develops, though, nitrifiers may elect to conserve their energy by limiting movement.
These `stress' conditions assuredly exist within nitrifying process reactors, involving reduced substrate presence, sub-optimal environments, and routine predation by higher life forms. In fact, the success of these reactors probably depends on this stress and the nitrifier's resultant shift toward surface attachment, as it is the only means by which an otherwise inevitable loss of free-swimming nitrifiers can be avoided.
Bacterial decay and death during prolonged resting or inactive periods occurs due to the natural onset of entropy. Aerobic bacteria may attempt to retard and prolong this tendency toward disorder through a process known as endogenous respiration, whereby a nominal energy flow is secured for limited maintenance purposes. This energy provides for resynthesis of critical metabolic material, as well as facilitating such activities as transport, motility, and pressure and heat control.
Our best estimate for the collective impacts of decay, endogenous metabolism, death, and predation on nitrifiers presently ranges from 0.05 to 0.12 days-1 . Correspondingly, these rates imply that resting, non-active nitrifiers will deteriorate at rates of 5 to 12% per day.
However, these values were typically inferred from heterotrophic bacteria rather than specifically measured for nitrifiers. Hence, conclusive data to clarify our understanding of nitrification-related decay and death is lacking.
Cell decay should certainly be expected with these organisms, but it is questionable whether their physiology would yield endogenous respiration activity during substrate-limited periods in a fashion comparable to that associated with heterotrophs. Indeed, energy procurement for these resting nitrifiers would probably depend more so upon external substrate availability than the mobilization and uptake of nitrogenous substrates found inside these cells. Resting nitrifying bacteria might consequently be expected to decay at a far lower pace than that which would be expected for heterotrophs. Correspondingly,indications within our contemporary literature that nitrifiers decay at a rate comparable to that of heterotrophs appear to be erroneous. In fact, unpublished values appear to be considerably lower (at about 0.01 to 0.02 days-1; Alleman, et al., 1991).
Slow decay and death rates for nitrifiers should, however, be considered a positive attribute. They can be shifted (e.g., taken off-line) into a dormant or resting state for extended periods with less concern about retaining their viability.
Nitrifiers need oxygen for normal metabolism. Every milligram of nitrogen passed through their full nitrification pathway (starting at ammonia and concluding at nitrate) requires approximately 4.5 mg of dissolved oxygen to scavenge electrons drawn from their nitrogenous substrates.
Should the available dissolved oxygen drop much below a few milligrams per liter, though, nitrifier metabolism will markedly slow down. However, even without access to any oxygen whatsoever, they can sustain some measure of respirative metabolism using nitrate or nitrite as an alternative electron acceptor (i.e. in lieu of oxygen). In short, they can, if necessary, both nitrify and denitrify, an incredible biochemical feat somewhat analogous to having their cake and eating it too.
Although both nitrifying bacteria appear to have this denitrifying capability, the biochemical balance between `nitritifiers' and `nitratifiers' may, itself, become disrupted by oxygen deprivation. Specifically, `nitritifiers' exposed to low oxygen conditions seemingly generate higher levels of intermediate hydroxylamine. In turn, the presence of this chemical will disrupt, and possibly terminate, the activity of their companion `nitratifiers,' resulting in a short-circuiting of the normal nitrification sequence.
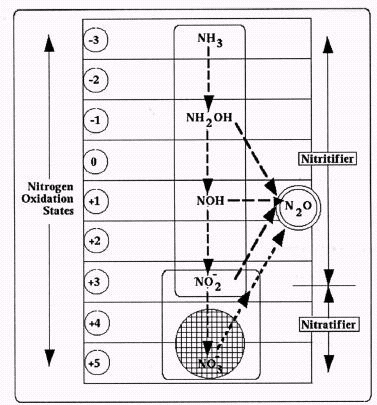
Having only discovered this denitrification capability within the last few years, our awareness and understanding of the nitrifier's denitrification capability is frankly tenuous. At this point, it appears that nitrifier denitrification often falls short of complete nitrate or nitrite reduction to dinitrogen gas. Instead, their `denitrification' behavior seemingly generates a gaseous nitrous oxide product, which in turn might well represent an important contributor to the greenhouse phenomenon.
Nitrifying bacteria prefer an alkaline environs, with optimal pH levels ranging from the mid-seven's to mid-eight's. For `nitritifiers,' their apparent preference for the high end of this range seemingly reinforces the hypothesis of their initial NH3-N substrate form.
Aside from the ambient pH itself, nitrifiers appear able to handle rather sizable dynamic transients in this parameter. However, after acclimation, it would be best for their activity if the pH were kept as consistent as possible. Concerns expressed over alkalinity stem principally from its correlation and impact on system pH. Alkalinity levels adequate to stop pH from dropping below the preferred alkaline range should be adequate.
Nitrifiers prefer moderate temperatures, ranging from 20 to 30°C. As temperature declines into the teens, their metabolic activity will decline. At temperature levels above 35°C, however, they enter a region of potential life-threatening stress, perhaps due to enzyme disruption. `Nitratifiers' specifically have an upper threshold of about 40°C, at which point their activity completely stops, while the permissible upper threshold for `nitritifiers' appears to be approximately 5°C higher. In either case, attention should be given to stabilizing reactor temperature, relative to avoiding extremes and short-term transients.
Nitrifiers have a sizable range of tolerable osmotic pressures, ranging from fresh to saline, depending on the particular genus form. Furthermore, many nitrifiers seem able to rapidly switch from one salt level to another with little impact on their activity. Within most nitrifying reactor systems, however, it is likely that they will become acclimated to an osmotic pressure which varies relatively little over time, even given the dynamics associated with makeup water addition. In turn, this parameter should not represent a particularly important concern.
Ammonia-oxidizing `nitritifiers' are sensitive to a region of the light spectrum known as near-shoulder' ultraviolet (Alleman, 1987). The exact cause of this negative impact is unknown, but may have some relationship with a superoxide radical produced in conjunction with stagnant membrane oxygen. For those `nitritifiers' affected by light, though, darkfield repair is possible, during which these bacteria rehabilitate over a period of hours in the presence of an available energy producing substrate.
The technical literature for nitrifiers has extensive references to the impact of specific chemicals on their efficacy, including, for example: thiourea, allythiourea, acetylene, N-Serve (Dow Chemical Corporation), heavy metals, metal chelators or binders (particularly those of copper), carbon disulfide, free ammonia, and free nitrous acid. Individuals interested in further details regarding this impacts should refer to the following citations: Blum and Speece (1991), Doster (1988), Pantea-Kiser (1987), and Sharma and Ahlert (1977).
As mentioned earlier, the `nitritifiers' employ an initial enzyme known as `ammonia monooxygenase' (AMO) which bears a surprising similarity to the methanotroph's `methane monooxygenase' (MMO). The importance behind this match stems from the fact that MMO is able to catalyze the oxidative breakdown of several hydrocarbons, including halogenated versions thereof.
Remarkably, recent studies of the AMO-bearing `nitritifiers' have shown that they too have this sort of ability, acting along a co-metabolism route in which their AMO becomes expended on an alternative substrate other than its intended ammonia form (Rasche, et al, 1990; and Hyman, et al., 1988).
In turn, `nitritifiers' may well have the ability to degrade industrial and hazardous wastes, at least to some extent. Unpublished estimates of this activity suggest that `nitritifiers' might be capable of degrading as much as one-tenth the weight of their daily nitrogen uptake in the form of co-metabolized hydrocarbons (Hyman, 1991).
Admittedly, aquaculture systems will not likely encounter or rely on this unusual capability. However, here again the recently derived insights on nitrifier behavior suggest that they are far more complex and robust that what they are usually given credit for.
An attached growth approach to nurturing nitrifier growth and retention would be recommended for aquaculture systems, in lieu of a suspended growth strategy. Stationary plastic packing material (nested sheets, random beads or saddles, etc.) represents a attractive media form, low in weight, high in specific surface area (e.g.,~ 30 ft2 per ft3 for many commercially-available media types), and chemically inert. Another option would be to use moving plastic media, such as rotating sheets or fluidized sand. Whatever the approach, attached cells can be retained in the reactor without having to deal with settling and recycle complications. This reactor configuration would be less prone to upset by hydraulic surges. However, the media must be consistently wetted to avoid spalling of dried biofilm. Furthermore, it would be highly recommended that some portion of this attached growth media be easily removable from the reactor to allow for visual inspection of the media and qualification of its related characteristics (e.g., film depth, film smell, etc.).
The principal operating parameter for these nitrifying units will be hydraulic loading. Unfortunately, recommended values vary considerably depending on media type. Working strictly on a `grass-roots' basis, though, one to two cubic foot of media could initially be used per 100 gal of tank volume, with a hydraulic loading rate of approximately one gallon per minute per square foot of cross-sectional media surface area.
Routine measurements should be made of the total NH4++NH3-N concentration in the bulk fluid. Simple `eye-ball' colorimetric analysis using Nesslers reagent (Standard Methods, 1975) would be adequate, with a desired result either having no color at all or a very, very faint yellow. Ideally, the bulk concentration should be consistently below 0.1 to 0.2 mg N/L; a pale yellow color on the Nesslers test would indicate an unacceptably high ammonia-nitrogen level, in the ~ 1 mg N/L range.
Routine measurements should be made of the NO2--N concentrations in the bulk fluid. Hach Chemical Company markets a convenient reagent `pillow' (NitriVer) which can be used for this test. Ideally, very little, if any, nitrite should be found in these systems. Elevated nitrite levels (above a 0.1 mg N/L concentration) will signal a metabolic upset within the full nitrification sequence.
Here again, routine measurements should be made of the NO3--N concentrations in the bulk fluid. Hach Chemical Company also sells reagent `pillows' (NitraVer) for nitrate. Nitrate will be expected to build up in the tanks, although diluted relative to the addition of dilution and makeup water.
Reactor temperature should be routinely monitored; values of ~ 20 --> 30°C are satisfactory. However, temperatures above 35°C must be avoided, as this level begins to approach a region of fatal stress for the `nitratifier.'
The pH level should be routinely monitored. Slightly alkaline values are preferable, within a desired range of ~ 7.5 to 8.2. pH levels above 9.0 to 9.5, or below 6, must be avoided since either extreme may harm the nitrifiers.
Alkalinity levels may be infrequently checked as a complement to the aforementioned pH measurements. Bicarbonate alkalinity levels of ~ 100 to 200 mg as CaCO3/L should be adequate.
Ideally, the nitrifiers should neither be exposed to sunlight or room light of a color other than red (e.g., darkroom lighting). When grown as an attached biofilm, though, considerable shading provided by bacterial layering will shelter the lower organisms from stressful light. Hence, this recommendation against light exposure is somewhat conservative.
The dissolved oxygen of water leaving the nitrification system should be routinely measured, and kept above 2 to 3 mg/L. Assumedly, the dissolved oxygen in the fish tanks will be much higher, and with reduced loading levels on the attached nitrifying bacteria, oxygen depletion should not be much of a concern.
Nitrifying biofilms (on media occasionally drawn from the nitrifying reactor) will tend to have a brown to orange-brown color, which intensifies into a reddish brown as the fraction of nitrifiers increases. Dark brown or blackish colors will denote problems with films that are deficient in oxygen or simply too thick.
Thick films (on media occasionally taken from the nitrifying reactor) much beyond a few millimeters will signify impending problems, particularly if the lower strata of the film appears dark black.
The biofilm should smell earth and musty, much like the bottom gravel found in a home aquarium. Sour or disagreeable smells, in particular, are tell-tale indicators of sub-optimal environmental conditions (i.e., probably septic).
If a microscope is readily available, routine visual inspections of the nitrifying biofilm would be recommended. Admittedly, it will be hard to pick out the nitrifiers. Instead, one should look for nuisance organisms which signify less than optimal environmental conditions inside the film. Filamentous bacteria, such as gliding Beggiatoa, are a prime example, as they tend to grow in low dissolved oxygen conditions unfavorable to the nitrifiers. Good films will normally include a diverse population of bacteria and higher life forms; in fact, it is likely that the nitrifiers will be far outnumbered within these mixed cultures.
Ammonia toxicity to fish contained within aquaculture systems can be reliably alleviated with biological nitrification, whereby excreted ammonia is successively oxidized to nitrite and nitrate. Given sufficient space and time, these nitrifiers can routinely scavenge ammonia from a recirculating flow passed over their attached media habitat. However, these microorganism have certain environmental preferences which must be routinely satisfied, including: elevated dissolved oxygen, neutral to slightly alkaline pH, and moderate temperature. Attention must also be given to their interaction with other bacteria, and specifically those who thrive on residual organic matter. These latter microbes can overwhelm the slower growing nitrifiers, overcrowding their niche and stifling their metabolism. These problems, however, can be minimized by providing additional media able to accommodate both groups by spreading their population over a greater, and effectively more viable, surface area.